


Triart Bio C18 is launched
Notice of discontinuation of Semi-Preparative Guard Cartridge column / Holder and introduction of alternative products
Change notification for the chromatographic data processors and inspection conditions (2019-1)
We are planning to replace the aging chromatographic data processors for inspection of YMC-Triart Diol-HILIC columns, starting from the production on July 22th, 2019. In order to pursue efficiency in production, we will also change some part of the inspection report format and inspection conditions of the columns.
It is confirmed that the change has no impact on product quality.
For more information, please click here.
Please do not hesitate to contact us if you have any questions or concerns.

Separation of chemically modified oligonucleotides using YMC-Triart Bio C4
Antisense DNA and siRNA have been widely used for gene silencing in basic research and in medicinal applications.
Effective delivery of the oligonucleotides into cells is important for clinical applications. Previous methods took several hours or more to deliver oligonucleotides to cytoplasm. However, oligonucleotides modified with low molecular weight disulfide units at their terminuses reached the cytoplasm 10 minutes after administration to cell culture 1).
In RP-HPLC using C18 columns, such disulfide modified oligonucleotides show poor peak shape due to too strong interaction between highly hydrophobic their disulfide units and C18 phase. On the other hand, YMC-Triart Bio C4 show a good peak shape because of its short alkyl chains and low hydrophobic interaction.
Separation of disulfide-modified oligonucleotides
Cell membrane permeable oligonucleotides
Oligonucleotides are negatively charged polymers, resulting in low cell membrane permeable efficiency. Various strategies are being pursued including chemical modification of oligonucleotides itself. Recently, it is reported that the disulfide-modified oligonucleotides are efficiently internalized into cytoplasm through disulfide exchange reactions with thiol groups on cell surface 1).
Separation of highly hydrophobic oligonucleotides
The chromatograms show the separation of phosphorothioate oligonucleotides modified with highly hydrophobic disulfide units. The target disulfide-modified oligonucleotides are not completely eluted from the C18 column due to the strong hydrophobic interaction. On the other hand, a good peak shape is obtained using YMC-Triart Bio C4, which has short alkyl chains, by the moderate interaction.
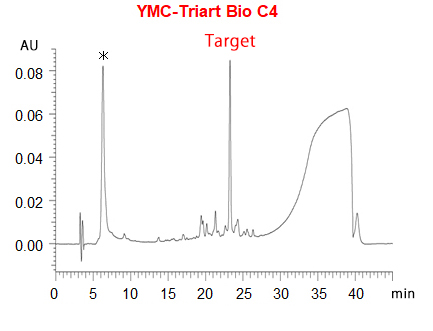
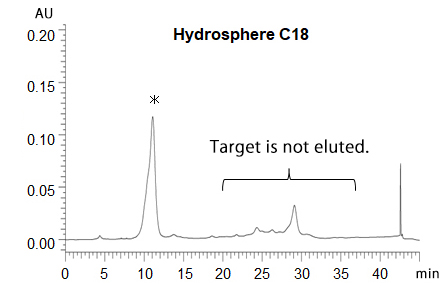
*phosphorothioate oligonucleotides without disulfide-unit

| Column | 5 µm, 250 X 4.6 mmI.D. A) 50 mM TEAA* (pH 7.0)/acetonitrile (95/5) B) acetonitrile 5-95%B (0-30 min), 95%B (30-35 min), 95-5%B (35-35.1 min), 5%B (35.1-45 min) |
|---|---|
| Flow rate | 1 mL/min |
| Temperature | 50°C |
| Detection | UV at 260 nm |
| Sample | crude reaction mixture |
* triethylammonium acetate
- Reference 1)
- Zhaome Shu et al. (2019) Disulfide-Unit Conjugation Enables Ultrafast Cytosolic Internalization of Antisense DNA and siRNA. Angew. Chem, 131, 6683-6687
Courtesy of Saki Kawaguchi, Chemistry Department, Nagoya University, Japan

Analysis of oxidized monoclonal antibodies by hydrophobic interaction chromatography
During manufacture and/or storage of biopharmaceuticals, variants with different properties from desired substances are produced by enzyme reactions or physicochemical interactions. Characterization of the variants is of great importance from the perspective of ensuring efficacy and safety of pharmaceutical products.
Oxidized mAb variants can be analyzed by hydrophobic interaction chromatography (HIC). On this page, we introduce the separation of mAb samples and their oxidized species using our HIC column, BioPro HIC BF.
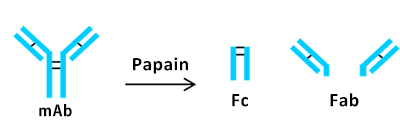
mAb oxidation with t-BHP treatment
tert-Butyl hydroperoxide (t-BHP) was used as a chemical oxidant to promote oxidation of methionine residues of NIST mAb.
Subsequently, papain was used for the preparation of two Fab fragments and one Fc fragment.
Analysis of oxidized mAb
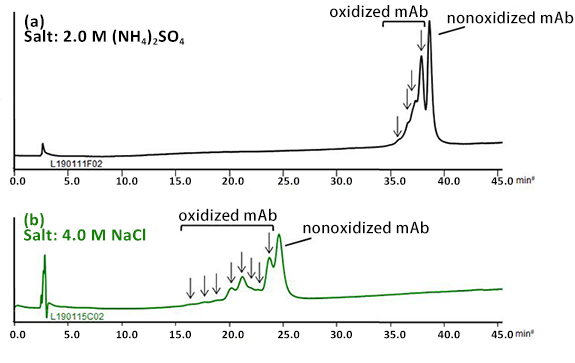
BioPro HIC BF column demonstrated the separation of oxidized mAb from the nonoxidized mAb using a low flow rate and shallow gradient slope.
In the chromatogram (a), earlier eluting four peaks were assumed to be derived from species that would have oxidized methionine residues on the mAb. The oxidation of sulfide side chains on methionine residues might result in conformational changes.
By using sodium chloride instead of ammonium sulfate, better resolution was achieved with a short analysis time (b).
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing salt B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) 40-80%B (0-40 min), 80%B (40-45 min) |
| Flow rate | 0.3 mL/min |
| Temperature | 25°C |
| Detection | UV at 280 nm |
| Injection | 5 µL (1.0 mg/mL) |
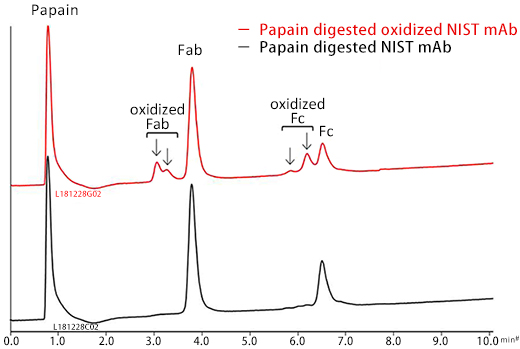
Analysis of papain digested oxidized mAb
Papain digested NIST mAb samples were analyzed. Fab and Fc peaks were observed on analysis of the nonoxidized mAb sample.
Peaks that would correspond to oxidized Fab and Fc were observed on the analysis of papain digested oxidized mAb sample. The oxidized species eluted earlier than the nonoxidized species. *
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing 2.0 M (NH4)2SO4 B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) 40-80%B (0-10 min) |
| Flow rate | 1.0 mL/min |
| Temperature | 25°C |
| Detection | UV at 280 nm |
| Injection | 5 µL (0.5 mg/mL) |
*Journal of Chromatography A, 2008, 1214, 81-89

The factor affecting separation of hydrophobic interaction chromatography
Hydrophobic interaction chromatography (HIC) is a technique used to separate proteins such as antibodies by hydrophobic interactions between proteins and stationary phase. The mobile phase is typically an aqueous buffer with high concentration. Proteins are adsorbed to the stationary phase at high concentration of salt, and elute in the order of increasing hydrophobicity by decreasing the salt concentration. Unlike reversed-phase, proteins can be separated without any denaturation, thereby maintaining its activity.
On this page, we introduce the factor affecting separation of HIC.
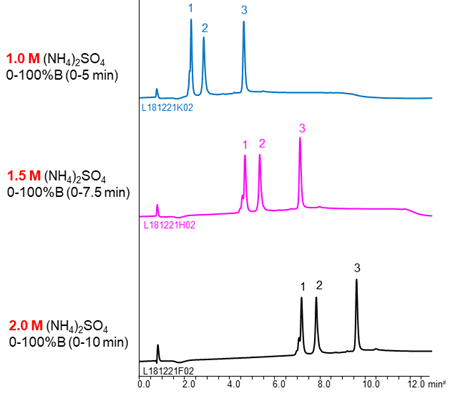
The effects of initial salt concentration and type of salt
The buffer containing (NH4)2SO4 is often used as mobile phase of HIC because (NH4)2SO4 has strong salting-out effect. The higher the concentration of initial (NH4)2SO4, the stronger retention of proteins, so a buffer with high salt concentration is effective for separation of the low hydrophobic proteins with weak retention.
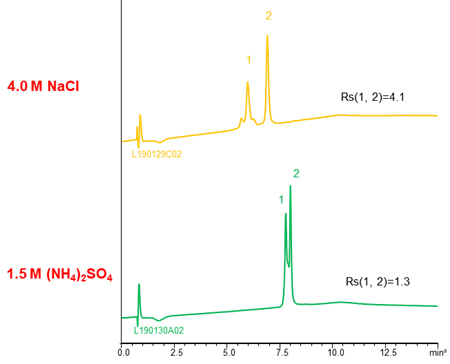
NaCl and CH3COONH4 are also used as salts. The separation selectivity vary with the type of salt in some cases (see chromatograms above), so changing the type of salt is also effective when the separation is poor. However, these salts are used very high concentration to gain retention comparable to (NH4)2SO4. It is need attention that precipitation of salts in the buffer and damage of LC system.
YMC’s column for hydrophobic interaction chromatography, BioPro HIC BF, is designed to high hydrophobicity of stationary phase. Therefore, it enables to analyze low hydrophobic proteins that can’t be retained using other commercial columns even in lower salt concentration buffer or low salting-out effect salts buffer.
The effects of initial salt concentration
- Adalimumab
- Trastuzumab
- Bevacizumab
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing salt B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) 0.2 M/min (The gradient slope is same.) |
| Flow rate | 1.0 mL/min |
| Temperature | 25°C |
| Detection | UV at 280 nm |
| Injection | 5 µL (0.5 mg/mL) |
The effects of type of salt
- Adalimumab
- NIST mAb
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing salt B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) 0-100%B (0-10 min), 100%B (10-15 min) |
| Flow rate | 1.0 mL/min |
| Temperature | 25°C |
| Detection | UV at 280 nm |
| Injection | 10 µL (0.25 mg/mL) |
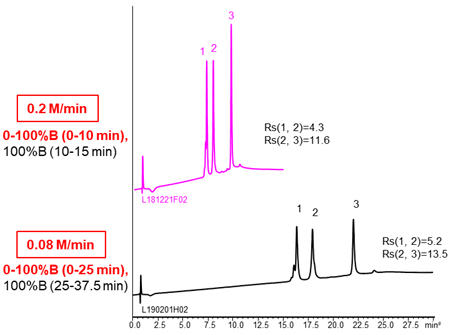
The effects of gradient slope
Using more shallow gradient make improve separation.
- Adalimumab
- Trastuzumab
- Bevacizumab
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing 2.0 M (NH4)2SO4 B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) |
| Flow rate | 1.0 mL/min |
| Temperature | 25°C |
| Detection | UV at 280 nm |
| Injection | 10 µL (0.5 mg/mL) |
The effects of temperature
In HIC mode, the higher temperature, the longer retention time of proteins. It assume that the hydrophobic area interacted with stationary phase become large by changing the structure of proteins with temperature increased, so the hydrophobic interaction become strong.
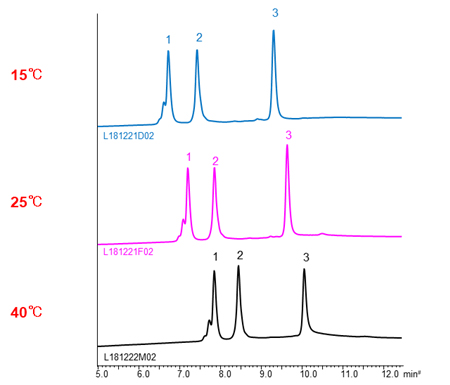
- Adalimumab
- Trastuzumab
- Bevacizumab
| Column | BioPro HIC BF 4 µm, 100 X 4.6 mmI.D. |
|---|---|
| Eluent | A) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) containing 2.0 M (NH4)2SO4 B) 100 mM NaH2PO4-Na2HPO4 (pH 7.0) 0-100%B (0-10 min), 100%B (10-15 min) |
| Flow rate | 1.0 mL/min |
| Detection | UV at 280 nm |
| Injection | 5 µL (0.5 mg/mL) |

LC-MS/MS analysis of sphingophospholipids using metal free column
Sphingolipids are major components forming biological membranes, and they serve as intracellular signaling molecules. It is important to measure the amount of these molecules in biological samples because it greatly influences on various metabolic diseases such as obesity, diabetes, and Alzheimer’s disease. However, on the LC analysis of sphingophospholipids, such as sphingosine-1-phosphate (S1P) and ceramide-1-phosphate (C1P), a phosphate group in those molecules causes significant peak tailing, and thus loss of sensitivity and reproducibility. Recently, the improved method using YMC-Triart C18 metal free column was reported by Dr. Gowda et al. 1)
Read more “LC-MS/MS analysis of sphingophospholipids using metal free column”



近期留言